Reprocessed batch shall be released in the market after the verification of data however the stability data of reprocessed batch shall be monitored for any significant change.īatch manufacturing record of Reprocessed batch shall be attached with the batch manufacturing record of the original batch. Reprocessed batch to be charge on stability shall be stamped on batch record charge on stability as per document control SOP (QA007). Reprocessed batches shall be charged for accelerated stability study. Suffix ‘P’ shall be added in the batch numbering of reprocessed batch in the existing batch numbering system. If such reprocessing is used for three batches, then the same process shall be considered as part of standard manufacturing process after consulting with R&D/Customer/RA and approval of Head-Quality Assurance. Reprocessing is normally a repetition of one of the previous step, already in a manufacturing process”. Reprocessing: It is defined as “Subjecting all or part of a batch or a lot of in-process stages or a finished product of single batch/lot to a previous step in the validated manufacturing process due to failure to meet predetermined specifications.Additional testing shall be carried out if required after justification for any in-processed stage or finished product of reprocessed, reworked and recoverable batch.Risk assessment shall be carried out before starting of the reworked, reprocessed or recoverable batch as per current version of Risk Assessment SOP, No.The batch shall be released only, if the quality of the final product is not affected, and the specifications are met.The reprocessing, reworking or recovery of products shall be carried out in case where there is any specific requirement and with prior approval of Manager-Quality Assurance/Customer/RA.Manager QA/ Authorized designee shall be responsible for approval and effective Implementation of the SOP. This SOP is applicable for handling of reprocessing, reworking and recovery of the drug products, at different stages of manufacturing, at the manufacturing facility.Īuthorized Designee of Quality Assurance shall be is responsible to prepare and review the SOP. When finished, all photos on the SD card or USB drive will display well on the digital photo frame.The purpose of this SOP is to lay down a procedure for reprocessing, reworking and recovery of the drug products. The photo reprocessing will take place.  
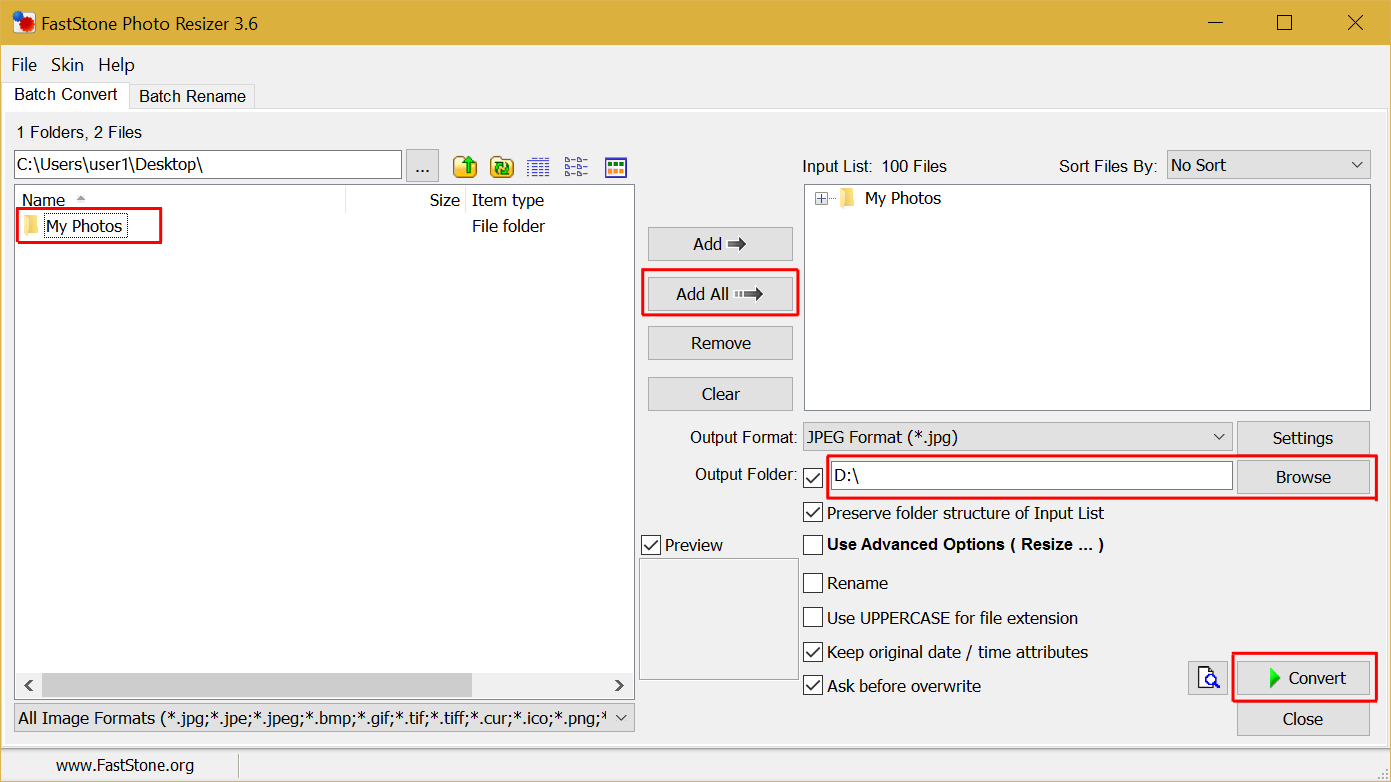
When finished, click on the “Convert” button. For our tutorial, our USB drive is drive D. Next, click on the “Output Folder” “Browse” button, and select your SD card or USB drive as the output folder. Highlight the folder with your photos in it, click on the “Add All” button to add the folder to the input list.Open FastStone Image Resizer and you should see a screen like below.Delete all photos from the SD card or USB drive – make sure you’ve copied them onto your computer.Copy all your photos into a folder on your computer desktop.First download and install the free software tool FastStone Image Resizer:.For Mac OS X users, click here for a tutorial that shows you how to do batch reprocessing using built-in tools. This tutorial shows you how to quickly fix this issue by saving all photos on to the SD card or USB drive in standard JPG format using a freely available software tool. When this happens it may be difficult to discover which photos are causing this issue. However, when there are corrupted or unsupported image files mixed in, customers may experience freezing, slow slideshow speed, or blurry pictures when viewing these photos. Micca digital photo frames display vast majority of photo images quickly and with excellent clarity.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
